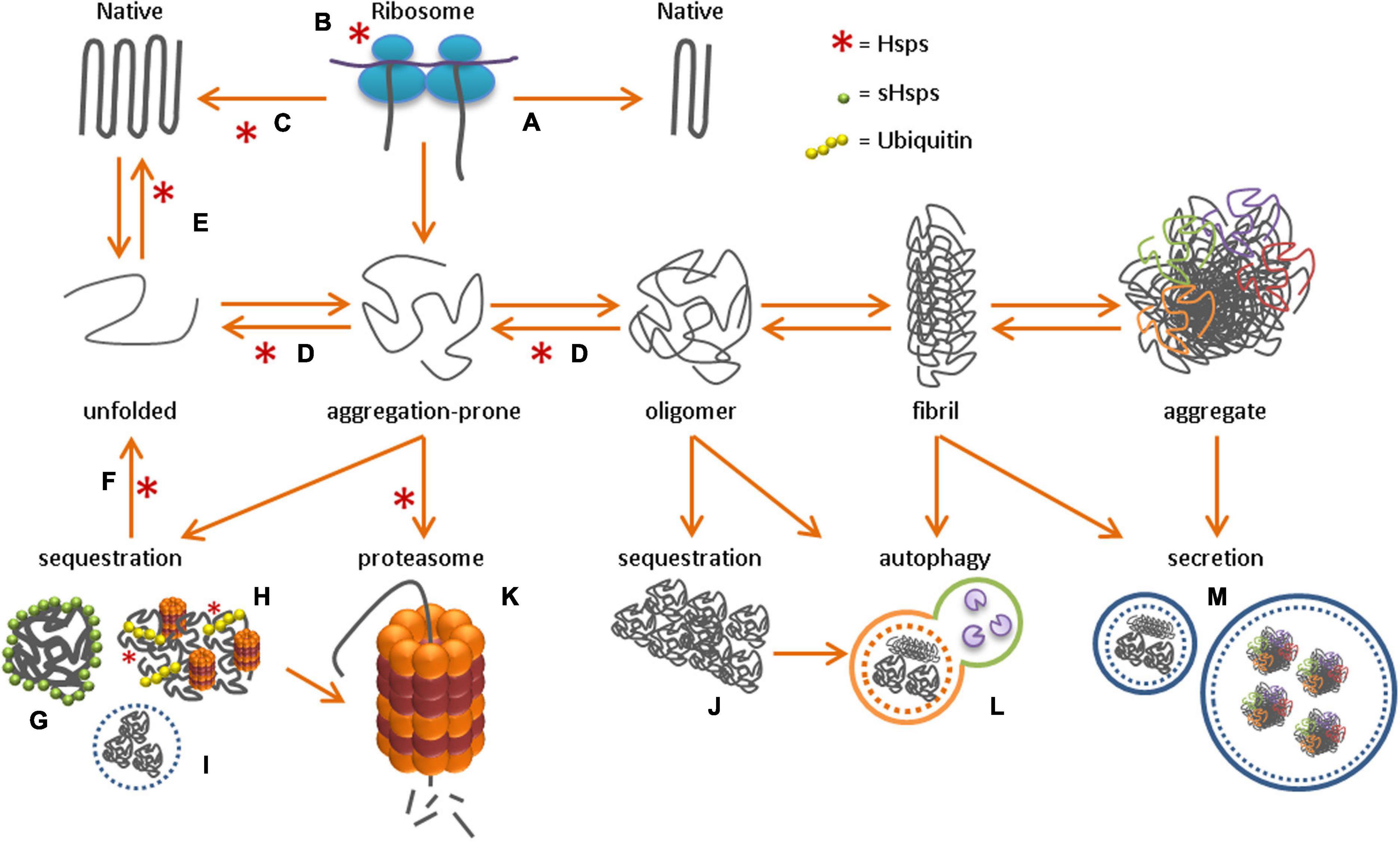
Probing the Origin of the Toxicity of Oligomeric Aggregates of α-Synuclein with Antibodies | ACS Chemical Biology
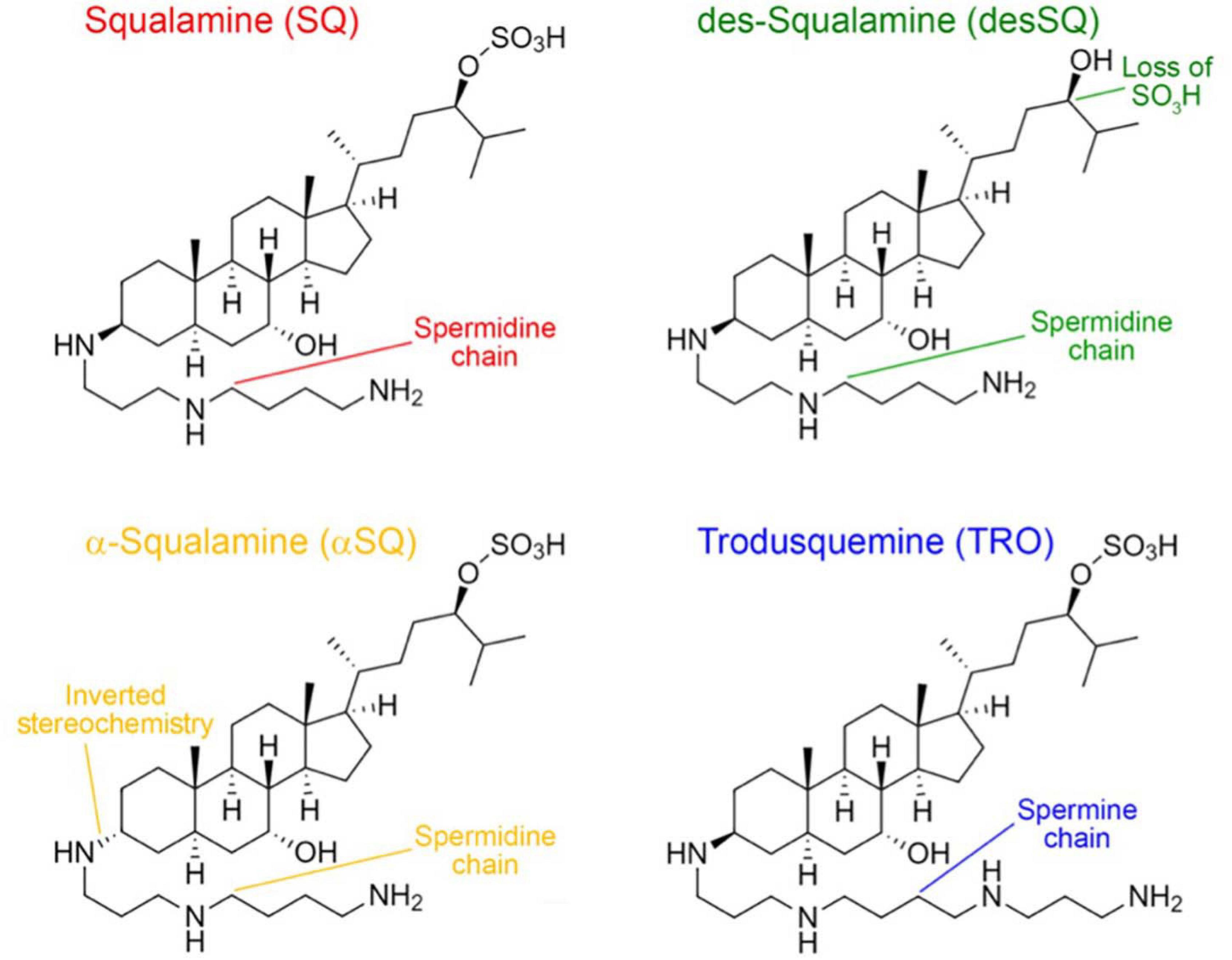
Frontiers | Squalamine and Its Derivatives Modulate the Aggregation of Amyloid-β and α-Synuclein and Suppress the Toxicity of Their Oligomers

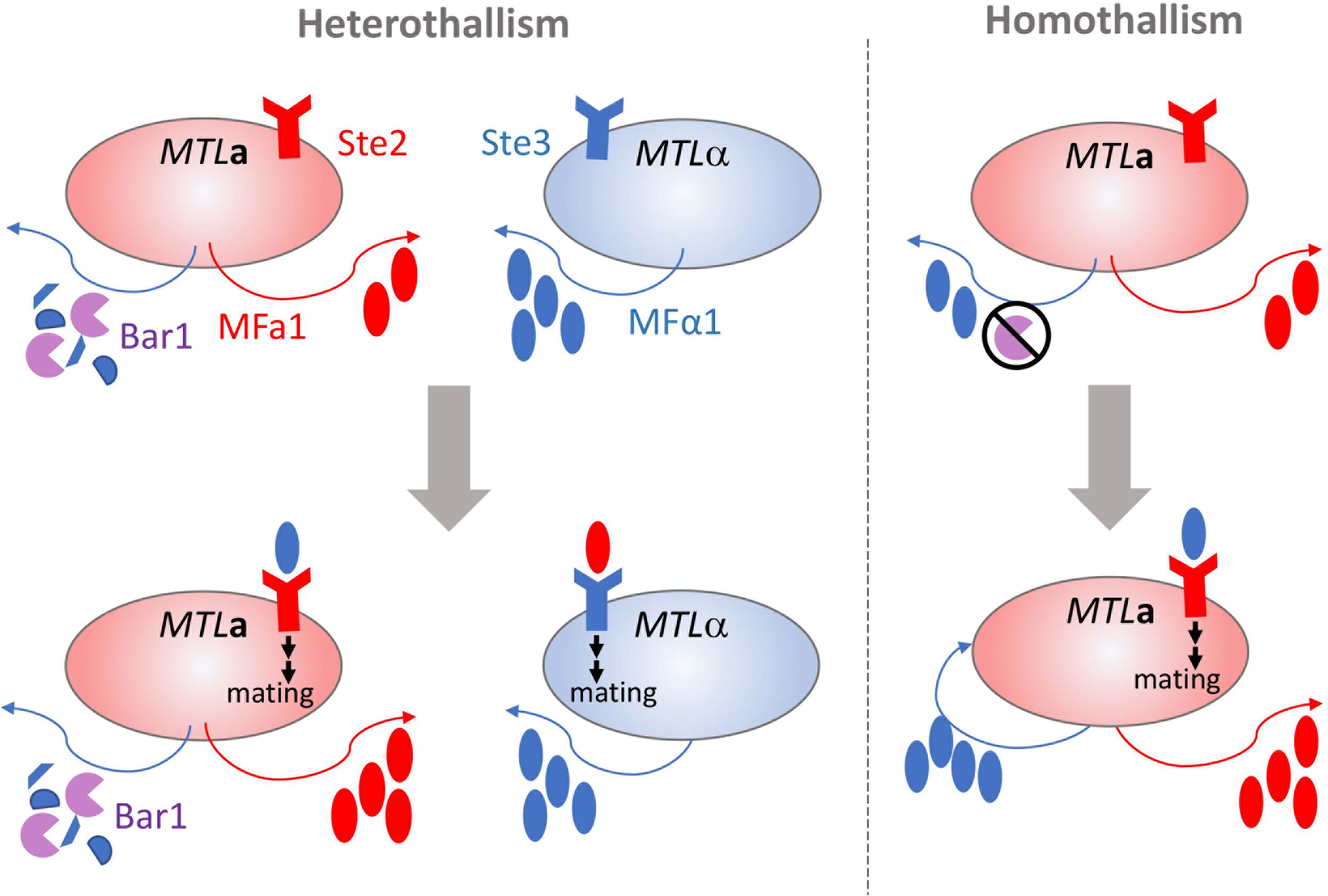
The cellular modifier MOAG‐4/SERF drives amyloid formation through charge complementation | The EMBO Journal

MOAG-4 promotes the aggregation of α-synuclein by competing with self-protective electrostatic interactions - ScienceDirect

A Brain-Permeable Aminosterol Regulates Cell Membranes to Mitigate the Toxicity of Diverse Pore-Forming Agents | ACS Chemical Neuroscience

Pawanrat CHALORAK | Instructor | PhD | Chulalongkorn University, Bangkok | Department of Radiological Technology | Research profile

MOAG-4 promotes the aggregation of α-synuclein by competing with self-protective electrostatic interactions - ScienceDirect

Akira KITAMURA | Senior Lecturer | Ph.D. | Hokkaido University, Sapporo | Hokudai | Faculty of Advanced Life Science | Research profile

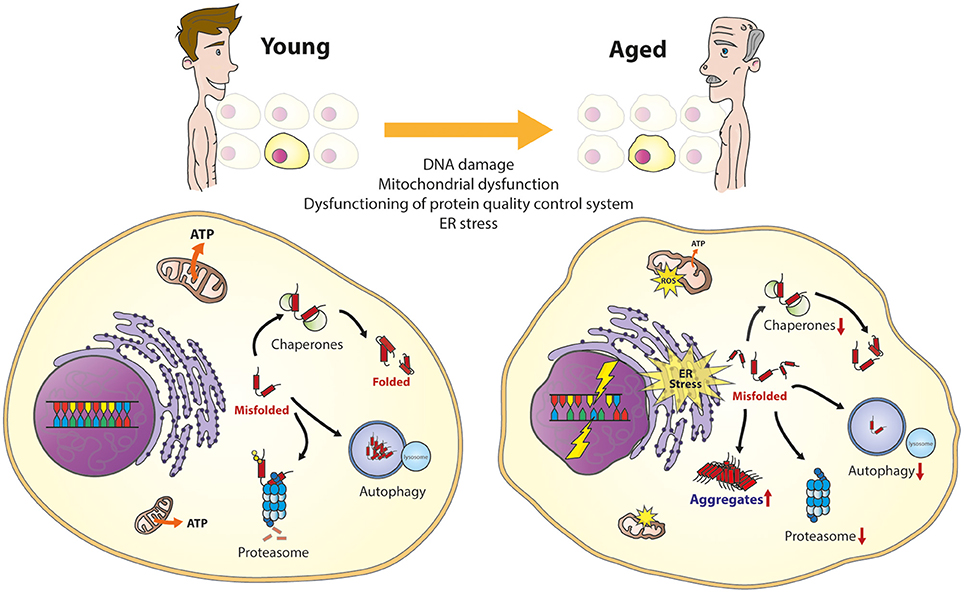
Deletion of SERF2 in mice delays embryonic development and alters amyloid deposit structure in the brain | Life Science Alliance

Delivery of Native Proteins into C. elegans Using a Transduction Protocol Based on Lipid Vesicles | Scientific Reports

Probing the Origin of the Toxicity of Oligomeric Aggregates of α-Synuclein with Antibodies | ACS Chemical Biology

Deletion of SERF2 in mice delays embryonic development and alters amyloid deposit structure in the brain | Life Science Alliance

The cellular modifier MOAG‐4/SERF drives amyloid formation through charge complementation | The EMBO Journal

The cellular modifier MOAG‐4/SERF drives amyloid formation through charge complementation | The EMBO Journal

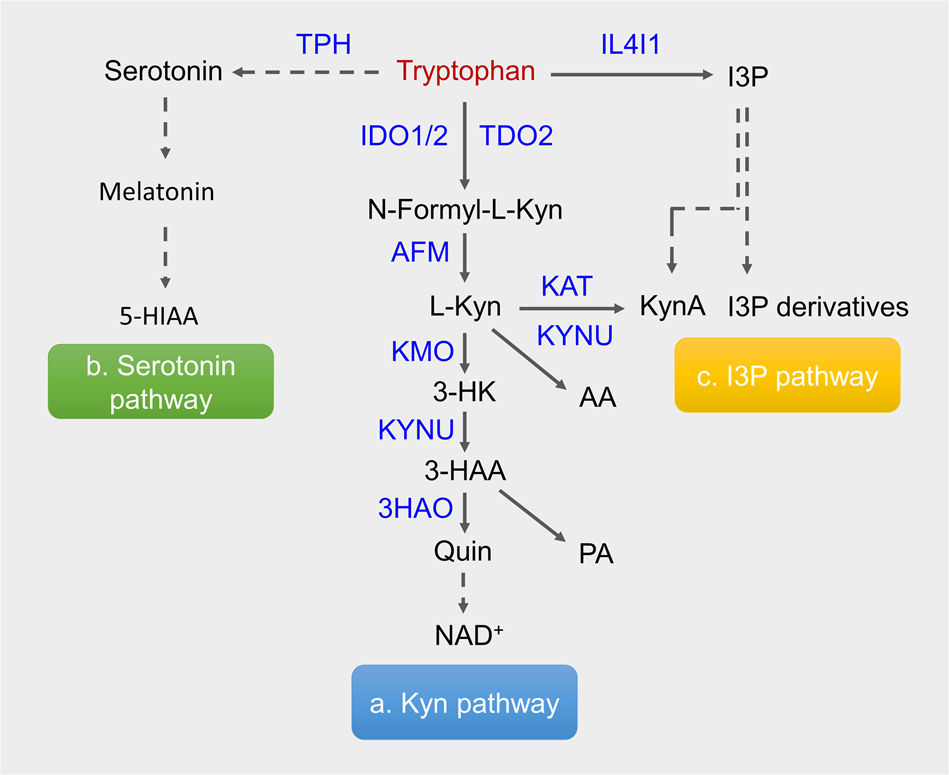
Crystal Structure and Mechanism of Tryptophan 2,3-Dioxygenase, a Heme Enzyme Involved in Tryptophan Catabolism and in Quinolinate Biosynthesis, | Biochemistry